
XP One brings standardized CSF infusion testing to the bedside, making the diagnostic pathway that underpins effective shunting accessible in more clinics, for more patients.
Visit our solutions page or send us a message at contact@x-pressure.com to learn more.
50 Years of Debate, Settled by One Trial
Shunt surgery for idiopathic normal pressure hydrocephalus (iNPH) has been performed since the 1960s. Yet for almost as long, a fundamental question has gone unanswered: does it truly work, or are patients simply responding to the expectation of improvement?
Without a large-scale, blinded, placebo-controlled trial, that question couldn’t be definitively answered, and clinicians were left making high-stakes decisions without the gold-standard evidence they deserved.
A Randomized Trial of Shunting for Idiopathic Normal-Pressure Hydrocephalus is the first large, international, randomized, double-blind, placebo-controlled study to test shunt surgery in iNPH.
Its conclusion: shunting works – and the evidence is overwhelming.
Trial Findings
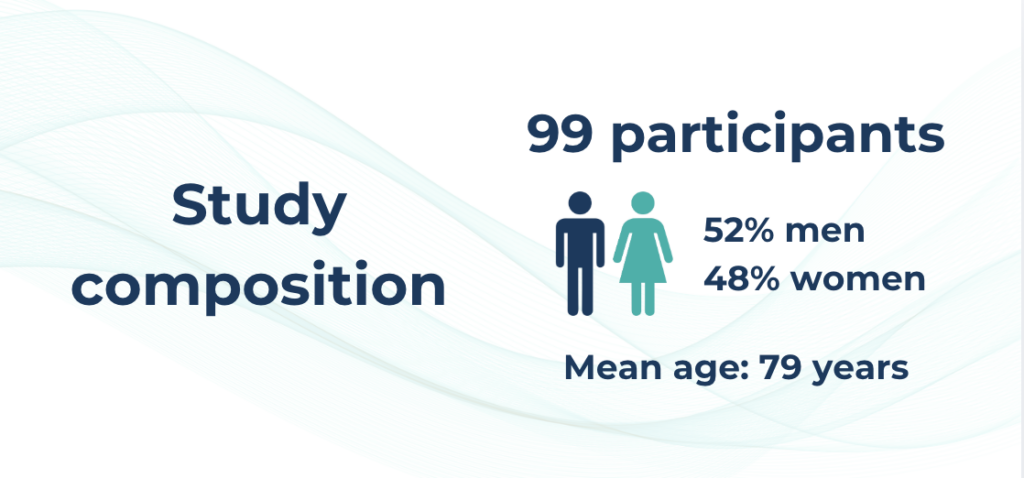
Ninety-nine patients across 17 centers in the United States, Canada, and Sweden were enrolled. All participants underwent shunt surgery, but were randomized to one of two groups: a functioning shunt (valve open at 110 mmH₂O) or a placebo shunt (valve set above 400 mmH₂O, effectively blocking CSF drainage). Neither patients nor clinicians knew which condition had been applied.
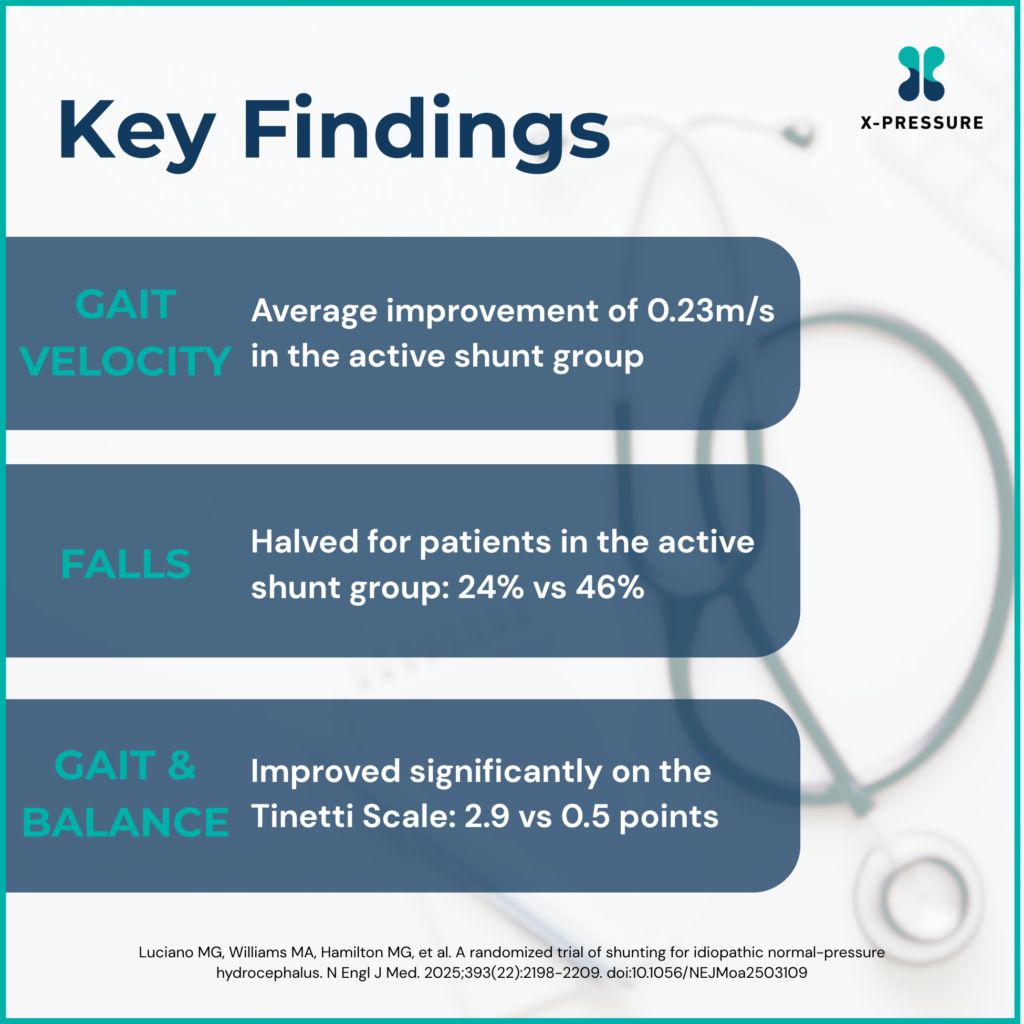
The primary outcome was change in gait velocity at three months – a direct, measurable indicator of the hallmark walking impairment in iNPH. Secondary outcomes included gait and balance (Tinetti Scale), cognition (Montreal Cognitive Assessment), and urinary incontinence.
The results were so compelling that the independent Data Safety Monitoring Board stopped enrollment early. This is an unusual step, reserved only for trials where the benefit becomes unambiguous before completion. All placebo-group participants were immediately directed to have their shunts opened.
A Serious Surgery, With Manageable Risks
The trial also addressed a key concern that has long fuelled caution: surgical risk in an elderly population.
Adverse events in the open-shunt group included subdural bleeding (12% vs 2% in the placebo group) and positional headaches (59% vs 28%). These numbers should not be minimized, but context matters. The majority of complications were successfully managed through non-invasive shunt valve adjustments, without further surgery.
When weighed against a near-halving of the fall rate, the risk-benefit calculation shifts substantially. Falls in elderly patients carry serious downstream consequences, including fractures, hospitalizations, loss of independence, and accelerated cognitive decline.
The PENS data makes a compelling case that, for appropriately selected patients, the risk profile of shunting is both acceptable and manageable.
The Detail That Changes Everything: How Patients Were Selected
The 80% responder rate in the open-shunt group is striking – and it didn’t happen by accident.
A positive response to a CSF drainage test before surgery was a prerequisite for enrollment in the PENS Trial. In other words, every patient had already been pre-screened for CSF-responsiveness before being randomized. The trial was not testing whether shunting works in any patient with suspected iNPH. It was testing whether it works in patients who had already demonstrated a likely response to CSF drainage.
This selection methodology aligns with decades of prior evidence underpinning the value of CSF infusion testing. The landmark Dutch NPH Study (Boon et al., 1997) demonstrated that elevated CSF outflow resistance predicts shunt response with a positive predictive value as high as 92–100% – findings that have formed the scientific foundation for modern infusion testing protocols.
The PENS Trial, then, is not just a vindication of shunting. It is an implicit endorsement of the diagnostic pathway that makes good patient selection possible.
Yet, researchers estimate that only an estimated 20% of patients who could benefit from shunt surgery are ever referred for evaluation. The PENS Trial makes a compelling case that this needs to change.
The Role of CSF Infusion Testing for Patient Selection
The PENS Trial confirms that shunting works, but it does not suggest that shunting works for everyone. The clinical imperative is identifying which patients are likely to respond – and doing so accurately, early, and at scale.
This is precisely the challenge that CSF infusion testing was designed to address. By measuring resistance to CSF outflow (Rout) in real time, it provides an objective, functional measure of whether a patient’s CSF dynamics are consistent with iNPH and whether they are, therefore, likely to benefit from surgical intervention. It is the bridge between clinical suspicion and evidence-based certainty.
X-Pressure’s mission is to ensure that CSF infusion testing, capable of assessing that responsiveness, will be available in more settings, to more clinicians, for more patients.
Visit our solutions page or contact us at contact@x-pressure.com to learn how XP One brings standardized CSF infusion testing to the bedside, making the diagnostic pathway that underpins effective shunting accessible in more clinics, for more patients.
